MSDS Cryopreserved Cells
Instructions HEK
5 Important Cell Culture Rules
Cell Apps Flyer Skin Cells
Cell Apps Poster Primary Cells
Cell Applications Inc Brochure
Description
Click thumbnails for more videos of HEK in action:
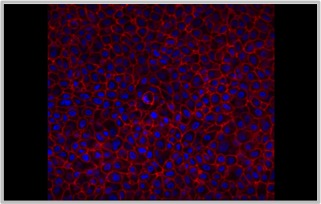
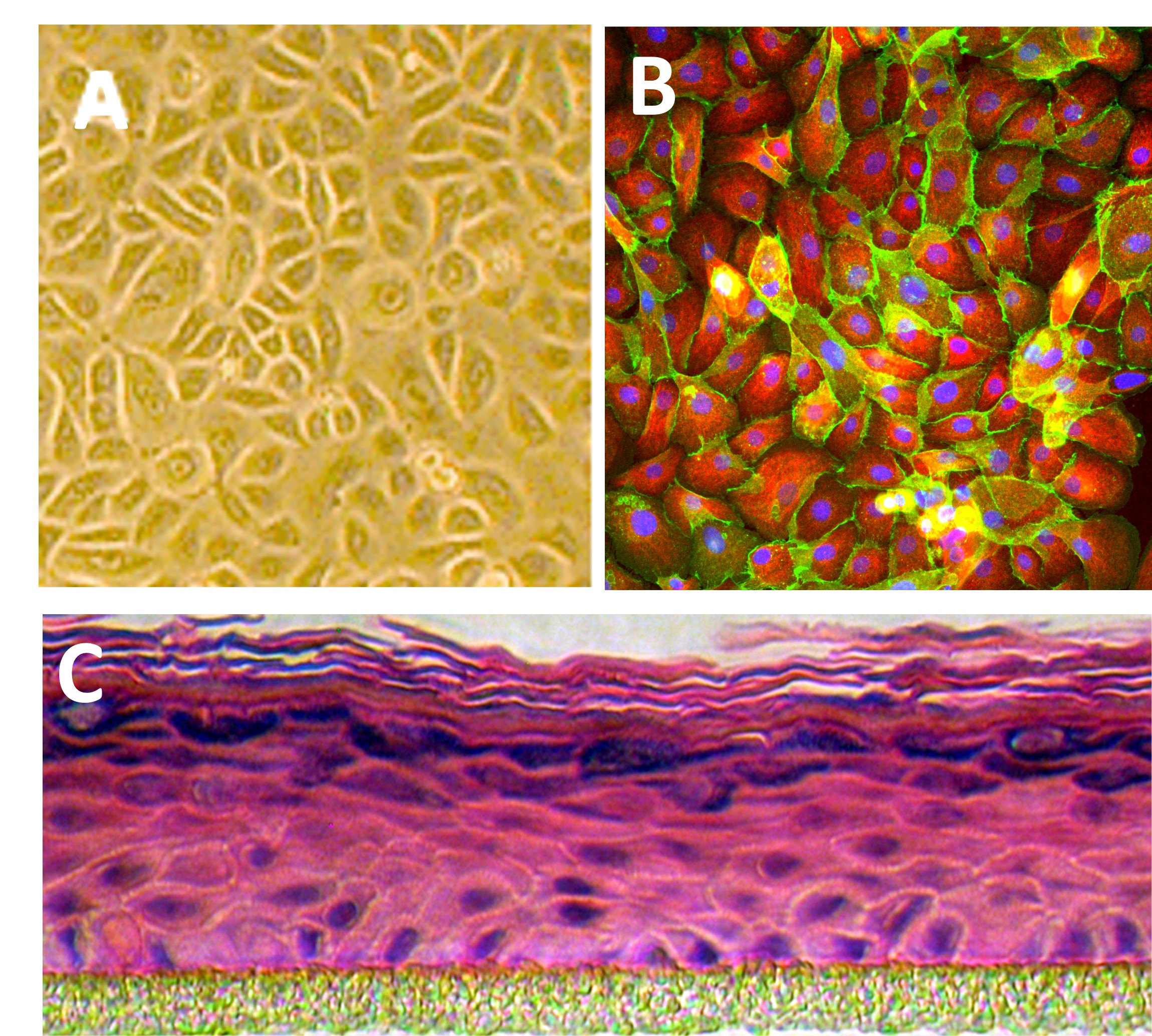
HEK from Cell Applications, Inc. have been utilized in numerous research publications, for example in basic research to investigate mechanisms of cell senescence and develop ways to extend cell life span. Others have used HEK to design a method to isolate proteins for use in basic research, and show higher relevance of primary keratinocytes when compared to key differences exhibited by immortalized keratinocyte cell lines. Pharmaceutical companies employ HEK to provide in vitro human skin models that assess effects of cosmetic formulations or anti-inflammatory properties of proposed anti-psoriasis drugs. Translational research on keratinocytes benefits from their ability to model the wound healing response, or help develop serum tests for patients with blistering diseases. HEK are also utilized to understand aggressiveness of cancer phenotypes, investigate compounds for potential topical melanoma treatments, and even develop siRNA therapy targeting oncogenes. In addition, HEK help unravel the oncogenic potential of HPV and test approaches for treating HPV lesions.
Details
Tissue | Normal healthy human foreskin or adult skin |
QC | No bacteria, yeast, fungi, mycoplasma, virus |
Bioassay | Attach, spread, proliferate in Growth Med |
Cryovial | 500,000 HEK (Primary culture) frozen in Basal Medium w/ 10% FBS, 10% DMSO |
Kit | Cryovial HEK (102-05), Epi-Vita Growth Medium (141-500a for adult; 141-500 for fetal/neonatal), Subcltr Rgnt Kit (090K) |
Proliferating | In Serum-Fr Gr Med, psg 1, flasks or plates |
| Doublings | At least 16 in keratinocyte media |
| Applications | Laboratory research use only (RUO). Not for human, clinical, diagnostic or veterinary use. |
Products
| Product | Size | CAT.# | Price | Quantity |
|---|---|---|---|---|
| Cryopreserved Human Epidermal Keratinocytes Total Kit, adult: 5x10^5 Cells (Adult), Medium & Subculture Reagents (See Details tab for specifics) | Size: 1 Kit | CAT.#: 102K-05a | Price: $661.00 | |
| Cryopreserved Epidermal Keratinocytes (HEK), adult: Frozen HEK (5x10^5) | Size: 1 Cryovial | CAT.#: 102-05a | Price: $475.00 | |
| Cryopreserved Human Epidermal Keratinocytes Total Kit, fetal: 5x10^5 Cells (Fetal), Medium & Subculture Reagents (See Details tab for specifics) | Size: 1 Kit | CAT.#: 102K-05f | Price: $675.00 | |
| Cryopreserved Epidermal Keratinocytes (HEK), fetal: Frozen HEK (5x10^5) | Size: 1 Cryovial | CAT.#: 102-05f | Price: $495.00 | |
| Cryopreserved Human Epidermal Keratinocytes Total Kit, neonatal: 5x10^5 Cells (Neonatal), Medium & Subculture Reagents (See Details tab for specifics) | Size: 1 Kit | CAT.#: 102K-05n | Price: $665.00 | |
| Cryopreserved Epidermal Keratinocytes (HEK), neonatal: Frozen HEK (5x10^5) | Size: 1 Cryovial | CAT.#: 102-05n | Price: $485.00 | |
| Proliferating Epidermal Keratinocytes (HEK), adult: Actively growing, dividing cells, in medium | Size: T-25 Flask | CAT.#: 103-25a | Price: $475.00 | |
| Proliferating Epidermal Keratinocytes (HEK), adult: Actively growing, dividing cells, in medium | Size: T-75 Flask | CAT.#: 103-75a | Price: $665.00 | |
| Proliferating Epidermal Keratinocytes (HEK), adult: Actively growing, dividing cells, in medium | Size: 24 Well | CAT.#: 103-24Wa | Price: $665.00 | |
| Proliferating Epidermal Keratinocytes (HEK), adult: Actively growing, dividing cells, in medium | Size: 96 Well | CAT.#: 103-96Wa | Price: $785.00 | |
| Proliferating Epidermal Keratinocytes (HEK), fetal: Actively growing, dividing cells, in medium | Size: T-25 Flask | CAT.#: 103-25f | Price: $495.00 | |
| Proliferating Epidermal Keratinocytes (HEK), fetal: Actively growing, dividing cells, in medium | Size: T-75 Flask | CAT.#: 103-75f | Price: $685.00 | |
| Proliferating Epidermal Keratinocytes (HEK), fetal: Actively growing, dividing cells, in medium | Size: 24 Well | CAT.#: 103-24Wf | Price: $685.00 | |
| Proliferating Epidermal Keratinocytes (HEK), fetal: Actively growing, dividing cells, in medium | Size: 96 Well | CAT.#: 103-96Wf | Price: $805.00 | |
| Proliferating Epidermal Keratinocytes (HEK), neonatal: Actively growing, dividing cells, in medium | Size: T-25 Flask | CAT.#: 103-25n | Price: $485.00 | |
| Proliferating Epidermal Keratinocytes (HEK), neonatal: Actively growing, dividing cells, in medium | Size: T-75 Flask | CAT.#: 103-75n | Price: $675.00 | |
| Proliferating Epidermal Keratinocytes (HEK), neonatal: Actively growing, dividing cells, in medium | Size: 24 Well | CAT.#: 103-24Wn | Price: $675.00 | |
| Proliferating Epidermal Keratinocytes (HEK), neonatal: Actively growing, dividing cells, in medium | Size: 96 Well | CAT.#: 103-96Wn | Price: $795.00 |
Related Products
| Product | Size | CAT.# | Price | Quantity |
|---|---|---|---|---|
| Human EpiVita Serum-Free Growth Medium for Adult Cells: All-in-one ready-to-use, standard Epithelial Cell Medium, for adult cells. Does not contain FBS. | Size: 500 ml | CAT.#: 141-500a | Price: $127.00 | |
| Human EpiVita Serum-Free Growth Medium Kit for Adult Cells: Basal medium & growth supplement sold together packaged separately, for adult cells. Does not contain FBS. | Size: Yields 500 ml | CAT.#: 141K-500a | Price: $140.00 | |
| Human EpiVita Serum-Free Growth Supplement for Adult Cells: Added to Basal Medium to create Growth Medium, for adult cells. Does not contain FBS. | Size: 5 ml | CAT.#: 141-GSa | Price: $64.00 | |
| Human EpiVita Serum-Free Growth Medium for Neonatal Cells: All-in-one ready-to-use, standard Epithelial Cell Medium, for neonatal and fetal cells. Does not contain FBS. | Size: 500 | CAT.#: 141-500 | Price: $121.00 | |
| Human EpiVita Serum-Free Growth Medium Kit for Neonatal Cells: Basal medium & growth supplement sold together packaged separately, for neonatal and fetal cells. Does not contain FBS. | Size: Yields 500 ml | CAT.#: 141K-500 | Price: $134.00 | |
| Human EpiVita Serum-Free Growth Supplement for Neonatal Cells: Added to Basal Medium to create Growth Medium, for neonatal and fetal cells. Does not contain FBS. | Size: 5 ml | CAT.#: 141-GS | Price: $59.00 | |
| Human EpiVita Basal Medium: Basal medium (contains no growth supplement). Add GS before use. | Size: 500 ml | CAT.#: 140-500 | Price: $96.00 |
Extended Family Products
| Product | Size | CAT.# | Price | Quantity |
|---|---|---|---|---|
| Human EpiVita Defined Growth Medium: All-in-one ready-to-use | Size: 500 | CAT.#: 141DGM-500 | Price: $121.00 | |
| Human EpiVita Defined Growth Medium Kit: Basal medium & growth supplement sold together packaged separately | Size: Yields 500 ml | CAT.#: 141DGMK-500 | Price: $133.00 | |
| Human EpiVita Basal Medium: Basal medium (contains no growth supplement). Add GS before use. | Size: 500 ml | CAT.#: 140-500 | Price: $96.00 | |
| Human EpiVita Defined Growth Supplement: Added to Basal Medium to create Growth Medium | Size: 5 ml | CAT.#: 141DGM-GS | Price: $61.00 | |
| Human Epidermal Cell Growth Medium: EpiVita Growth Medium without calcium. Calcium must be added to support cell growth. | Size: 500 ml | CAT.#: 133-500 | Price: $129.00 | |
| Human Epidermal Cell Growth Medium Kit: EpiVita Growth Medium Kit without calcium. Calcium must be added to support cell growth. | Size: yields 500 ml | CAT.#: 133K-500 | Price: $139.00 | |
| Human Epidermal Cell Growth Medium Kit: EpiVita Growth Medium Kit for adult cells without calcium. Calcium must be added to support cell growth. | Size: Yields 500 ml | CAT.#: 133K-500a | Price: $146.00 | |
| Human Epidermal Cell Basal Medium: Basal medium without calcium | Size: 500 ml | CAT.#: 132-500 | Price: $91.00 | |
| Human Epidermal Cell Growth Supplement: EpiVita Growth Supplement without calcium. Calcium must be added to support cell growth. | Size: 5 ml (3 parts) | CAT.#: 133-GS | Price: $66.00 | |
| 3-D Skin Model: HEK differentiated into stratified squamous epithelium on PCF sitting inserts. Available only in the US. Approximate turnaround time 3-5 weeks. | Size: 12 Sitting Inserts | CAT.#: 102-3D-12 | Price: $672.00 | |
| 3-D Skin Model (Not available for international orders): HEK differentiated into stratified squamous epithelium on PCF sitting inserts. Available only in the US. Approximate turnaround time 3-5 weeks. | Size: 24 Sitting Inserts | CAT.#: 102-3D-24 | Price: $1,276.00 | |
| 3-D Skin Model: Cells, Media, Reagents & Inserts (See Details tab for specifics) | Size: 1 Kit | CAT.#: 102K-3D | Price: $640.00 | |
| Polyclonal CXCR1 Rabbit Antibody: Polyclonal CXCR1 Rabbit Antibody | Size: 100 ul | CAT.#: CA0755 | Price: $375.00 | |
| Polyclonal CXCR2 Rabbit Antibody: Polyclonal CXCR2 Rabbit Antibody | Size: 100 ul | CAT.#: CA0757 | Price: $375.00 | |
| Human Epidermal Growth Factor (EGF): Human Epidermal Growth Factor | Size: 100 ug | CAT.#: RP1026-100 | Price: $86.00 | |
| Human Epidermal Growth Factor (EGF): Human Epidermal Growth Factor | Size: 500 ug | CAT.#: RP1026-500 | Price: $194.00 | |
| Human Epidermal Growth Factor (EGF): Human Epidermal Growth Factor | Size: 1000 ug | CAT.#: RP1026-1000 | Price: $264.00 | |
| Epidermal Keratinocyte RNA (HEK RNA), Adult: Total RNA prepared from Human Epidermal Keratinocytes, adult | Size: 10 ug | CAT.#: 102-R10a | Price: $398.00 | |
| Epidermal Keratinocyte RNA (HEK RNA), Adult: Total RNA prepared from Human Epidermal Keratinocytes, adult | Size: 25 ug | CAT.#: 102-R25a | Price: $796.00 | |
| Epidermal Keratinocyte RNA (HEK RNA), Fetal: Total RNA prepared from Human Epidermal Keratinocytes, fetal | Size: 10 ug | CAT.#: 102-R10f | Price: $398.00 | |
| Epidermal Keratinocyte RNA (HEK RNA), Fetal: Total RNA prepared from Human Epidermal Keratinocytes, fetal | Size: 25 ug | CAT.#: 102-R25f | Price: $796.00 | |
| Epidermal Keratinocyte RNA (HEK RNA), Neonatal: Total RNA prepared from Human Epidermal Keratinocytes, neonatal | Size: 10 ug | CAT.#: 102-R10n | Price: $398.00 | |
| Epidermal Keratinocyte RNA (HEK RNA), Neonatal: Total RNA prepared from Human Epidermal Keratinocytes, neonatal | Size: 25 ug | CAT.#: 102-R25n | Price: $796.00 | |
| Subculture Reagent Kit: 100 ml each of HBSS, Trypsin/EDTA & Trypsin Neutralizing Solution | Size: 1 Kit | CAT.#: 090K | Price: $69.00 | |
| Human EGF, Animal-Free: Human Epidermal Growth Factor, Animal-Free | Size: 100 ug | CAT.#: RP1026AF-100 | Price: $95.00 | |
| Human EGF, Animal-Free: Human Epidermal Growth Factor, Animal-Free | Size: 500 ug | CAT.#: RP1026AF-500 | Price: $213.00 | |
| Human EGF, Animal-Free: Human Epidermal Growth Factor, Animal-Free | Size: 1000 ug | CAT.#: RP1026AF-1000 | Price: $290.00 | |
| Human EpiVita Serum-Free Growth Medium for Neonatal Cells without Antibiotics: All-in-one ready-to-use, standard Epithelial Cell Medium, for neonatal and fetal cells. Does not contain FBS or Antibiotics. | Size: 500 ml | CAT.#: 141A-500 | Price: $134.00 | |
| Human EpiVita Serum-Free Growth Medium Kit for Adult Cells, without Antibiotics: Basal medium & growth supplement sold together packaged separately, for adult cells. Does not contain FBS or Antibiotics. | Size: Yields 500 ml | CAT.#: 141AK-500a | Price: $152.00 | |
| Human EpiVita Serum-Free Growth Medium for Adult Cells, without Antibiotics: All-in-one ready-to-use, standard Epithelial Cell Medium, for adult cells. Does not contain FBS or Antibiotics. | Size: 500 ml | CAT.#: 141A-500a | Price: $140.00 | |
| Human EpiVita Serum-Free Growth Medium Kit for Neonatal Cells, without Antibiotics: Basal medium & growth supplement sold together packaged separately, for neonatal and fetal cells. Does not contain FBS or Antibiotics. | Size: Yields 500 ml | CAT.#: 141AK-500 | Price: $145.00 |
Resources/Documents
Citations
Publications
2016
Tanihara, M., K. Takaichi, M. Maeda, T. Mitsui, K. Yamamoto and A. Hirano. 2016. Retinol-modified collagen, method for producing same, and external composition for skin containing same. Patent US 9238688 B2.
2015
Jie, L., R.Huang, W. Sun, S. Wei, Y. Chu, Q. Huang, and H. Du. 2015. Role of cysteine‑rich angiogenic inducer 61 in fibroblast‑like synovial cell proliferation and invasion in rheumatoid arthritis. Molecular Medicine Reports, 11:917-923.
Hsiao, Y., W. Lai, S. Wu, C. Tsai, S. Tang, J. Chung, and J. Yang. 2015. Triggering Apoptotic Death of Human Epidermal Keratinocytes by Malic Acid: Involvement of Endoplasmic Reticulum Stress- and Mitochondria-Dependent Signaling Pathways. Toxins, 7:81-96.
Kubat, N., J. Moffett, and L. Fray. 2015. Effect of pulsed electromagnetic field treatment on programmed resolution of inflammation pathway markers in human cells in culture. J Inflamm Res, 8:59-69.
Togtema, M., R. Jackson, C. Richard, S. Niccoli and I. Zehbe. 2015. The human papillomavirus 16 European-T350G E6 variant can immortalize but not transform keratinocytes in the absence of E7. Virology, 485:274-282.
Yan, Y., M. Furumura, T. Gouya, A. Iwanaga, K. Teye, S. Numata, T. Karashima, X. Li, and T. Hashimoto. 2015. Shikonin Promotes Skin Cell Proliferation and Inhibits Nuclear Factor-κB Translocation via Proteasome Inhibition In Vitro. Chinese Medical Journal, 128:2228-2233.
2014
Hirako, Y., Y. Yonemoto, T. Yamauchi, Y. Nishizawa, Y. Kawamoto, and K. Owaribe. 2014. Isolation of a hemidesmosome-rich fraction from a human squamous cell carcinoma cell line. Experimental cell research. 324:172-82.
Inoue, Y. S. Hasegawa, S. Ban, T. Yamada, Y. Date, H. Mizutani, S. Nakata, M. Tanaka, and N. Hirashima. 2014. ZIP2 Protein, a Zinc Transporter, Is Associated with Keratinocyte Differentiation. JBC, 289:21451-21462.
Kumar, G., H. Man, R. Chen, G. Muller, and A. Frank. 2014. Phenethylsulfone isoindoline derivatives and their use. Patent US 8853175 B2. Ohsumi, K., M. Watanabe, and A. Fujie. 2014. AS2077715 is a selective inhibitor of fungal mitochondrial cytochrome bc1 complex. The Journal of Antibiotics, 713–716.
Schafer, P. A. Frank, H. Man, and S. Shankar. 2014. (Methylsulfonyl) ethyl benzene isoindoline derivatives and their pharmaceutical uses. Patent US 8716252 B2.
2012
DiNatale, B.C., K. Smith, K. John, G. Krishnegowda, S.G. Amin, and G.H. Perdew. 2012. Ah receptor antagonism represses head and neck tumor cell aggressive phenotype. Molecular Cancer Research. 10:1369-1379.
Egawa, N., T. Nakahara, S.-i. Ohno, M. Narisawa-Saito, T. Yugawa, M. Fujita, K. Yamato, Y. Natori, and T. Kiyono. 2012. The E1 protein of human papillomavirus type 16 is dispensable for maintenance replication of the viral genome. Journal of virology. 86:3276-3283.
Garbe, J. 2012. Vulnerability of Normal Human Mammary Epithelial Cells to Oncogenic Transformation. Award Number: W81XWH-07-1-0643.
Moffett, J., L.M. Fray, and N.J. Kubat. 2012. Activation of endogenous opioid gene expression in human keratinocytes and fibroblasts by pulsed radiofrequency energy fields. Journal of pain research. 5:347.
Niccoli, S. 2012. The carcinogenic properties of naturally occurring human papillomavirus 16 E6 oncogene variants. Lakehead University, PhD dissertation.
Niccoli, S., S. Abraham, C. Richard, and I. Zehbe. 2012. The Asian-American E6 variant protein of human papillomavirus 16 alone is sufficient to promote immortalization, transformation, and migration of primary human foreskin keratinocytes. Journal of virology. 86:12384-12396.
2011
Akaza, N., H. Akamatsu, M. Kishi, H. Mizutani, S. Nakata, and K. Matsunaga. 2011. Normal human epidermal keratinocytes react differently than HaCaT keratinocyte cell line on exposure to Propionibacterium acnes. The Journal of Dermatology. 38:499-502.
Moffett, J., N.J. Kubat, N.E. Griffin, M.C. Ritz, and F.R. George. 2011. Pulsed radio frequency energy field treatment of cells in culture: Increased expression of genes involved in angiogenesis and tissue remodeling during wound healing. The Journal of Diabetic Foot Complications. 3:30-39.
Tanihara, M., K. Takaichi, M. Maeda, T. Mitsui, K. Yamamoto, and A. Hirano. 2011. Retinol-modified collagen, method for producing same, and external composition for skin containing same. Patent Application US 20130116189 A1.
Yamato, K., N. Egawa, S. Endo, K. Ui-Tei, T. Yamada, K. Saigo, I. Hyodo, T. Kiyono, and I. Nakagawa. 2011. Enhanced specificity of HPV16 E6E7 siRNA by RNA–DNA chimera modification. Cancer gene therapy. 18:587-597.
Zehbe, I., C. Richard, K.F. Lee, M. Campbell, L. Hampson, and I.N. Hampson. 2011. Lopinavir shows greater specificity than zinc finger ejecting compounds as a potential treatment for human papillomavirus-related lesions. Antiviral research. 91:161-166.
2010
Del Carmen Velazquez Pereda, M., G. De Campos Dieamant, S. Eberlin, R.M. Werka, D. Colombi, M.L. De Souza Queiroz, and L.C. Di Stasi. 2010. Expression of differential genes involved in the maintenance of water balance in human skin by Piptadenia colubrina extract. Journal of Cosmetic Dermatology. 9:35-43.
Flynn, R., J.M. Buckler, C. Tang, F. Kim, and D.A. Dichek. 2010. Helper-dependent Adenoviral Vectors are Superior In Vitro to First-generation Vectors for Endothelial Cell-targeted Gene Therapy. Molecular therapy : the journal of the American Society of Gene Therapy. 18:2121-2129.
Moffett, J., N. Griffin, M. Ritza, and F. George. 2010. Pulsed radio frequency energy field treatment of cells in culture results in increased expression of genes involved in the inflammation phase of lower extremity diabetic wound healing. J Diabetic Foot Complications, 2:57-64.
Richard, C., C. Lanner, S. Naryzhny, L. Sherman, H. Lee, P. Lambert, and I. Zehbe. 2010. The immortalizing and transforming ability of two common human papillomavirus 16 E6 variants with different prevalences in cervical cancer. Oncogene. 29:3435-3445.
Schafer, P., A. Parton, A. Gandhi, L. Capone, M. Adams, L. Wu, J. Bartlett, M. Loveland, A. Gilhar, and Y.F. Cheung. 2010. Apremilast, a cAMP phosphodiesterase‐4 inhibitor, demonstrates anti‐inflammatory activity in vitro and in a model of psoriasis. British journal of pharmacology. 159:842-855.
Thuangtong, R., J.J. Bentow, K. Knopp, N.A. Mahmood, N.E. David, and M.S. Kolodney. 2010. Tissue‐Selective Effects of Injected Deoxycholate. Dermatologic Surgery. 36:899-908.
Vrba, L., T.J. Jensen, J.C. Garbe, R.L. Heimark, A.E. Cress, S. Dickinson, M.R. Stampfer, and B.W. Futscher. 2010. Role for DNA Methylation in the Regulation of miR-200c and miR-141 Expression in Normal and Cancer Cells. PloS one. 5:e8697.
2009
Eberlin, S., M. Del Carmen Velazquez Pereda, G. De Campos Dieamant, C. Nogueira, R.M. Werka, and M.L. De Souza Queiroz. 2009. Effects of a Brazilian herbal compound as a cosmetic eyecare for periorbital hyperchromia (“dark circles”). Journal of Cosmetic Dermatology. 8:127-135.
Zhang, L.-S., W.M. Petroll, H.J. Greyner, and M.E. Mummert. 2009. Development of a hyaluronan bioconjugate for the topical treatment of melanoma. Journal of dermatological science. 55:56.
2008
Dieamant, G.d.C., M.D.C. Velazquez Pereda, S. Eberlin, C. Nogueira, R.M. Werka, and M.L.d.S. Queiroz. 2008. Neuroimmunomodulatory compound for sensitive skin care: in vitro and clinical assessment. Journal of Cosmetic Dermatology. 7:112-119.
Shafikhani, S.H., K. Mostov, and J. Engel. 2008. Focal adhesion components are essential for mammalian cell cytokinesis. Cell cycle. 7:2868-2876.
2007
Haga, K., S.-i. Ohno, T. Yugawa, M. Narisawa-Saito, M. Fujita, M. Sakamoto, D.A. Galloway, and T. Kiyono. 2007. Efficient immortalization of primary human cells by p16INK4a-specific short hairpin RNA or Bmi-1, combined with introduction of hTERT. Cancer science. 98:147-154.
Narisawa-Saito, M., K. Handa, T. Ygawa, S. Ohno, M. Fujita, and T. Kiyono. 2007. HPV16 E6-mediated stabilization of ErbB2 in neoplastic transformation of human cervical keratinocytes. Oncogene, 26;2988-2996.
Perrais, M., X. Chen, M. Perez-Moreno, and B.M. Gumbiner. 2007. E-Cadherin Homophilic Ligation Inhibits Cell Growth and Epidermal Growth Factor Receptor Signaling Independently of Other Cell Interactions. Molecular biology of the cell. 18:2013-2025.
Tao, H., A.J. Berno, D.R. Cox, and K.A. Frazer. 2007. In Vitro Human Keratinocyte Migration Rates Are Associated with SNPs in the KRT1 Interval. PloS one. 2:e697.
Yugawa, T., K. Handa, M. Narisawa-Saito, S.-i. Ohno, M. Fujita, and T. Kiyono. 2007. Regulation of Notch1 Gene Expression by p53 in Epithelial Cells. Molecular and cellular biology. 27:3732-3742.
2006
Baron, U., I. Turbachova, A. Hellwag, F. Eckardt, K. Berlin, U. Hoffmüller, P. Gardina, and S. Olek. 2006. DNA Methylation Analysis as a Tool for Cell Typing. Epigenetics 1:55-60.
Shafikhani, S.H., and J. Engel. 2006. Pseudomonas aeruginosa type III-secreted toxin ExoT inhibits host-cell division by targeting cytokinesis at multiple steps. Proceedings of the National Academy of Sciences. 103:15605-15610.
2004
George, F., and J. Moffett. 2004. Using electromagnet energy to propagate and stimulate cell growth; regenerative medicine; wound healing agents; tissue engineering and treatment of skin disorders. Patent US 20050059153 A1.
2003
Hatakeyama, S., Y.Z. Ma, N. Miura, S. Abe, T. Kameda, K. Sakamoto, and T. Sugiyama. 2003. Production of monoclonal antibodies recognizing human hair follicle keratinocytes. Hybridoma and hybridomics. 22:127-130.
Kameda, T. 2003. MAb-sDP. Hybridoma and Hybridomics. 22:191-191.
Kameda, T., 2003. MAb-8G2. Hybridoma and Hybridomics. 22:192-192.
Tsuji, H., K. Okamoto, Y. Matsuzaka, H. Iizuka, G. Tamiya, and H. Inoko. 2003. SLURP-2, a novel member of the human Ly-6 superfamily that is up-regulated in psoriasis vulgaris. Genomics. 81:26-33.
2002
Huh, W.-K., T. Oono, Y. Shirafuji, H. Akiyama, J. Arata, M. Sakaguchi, N.-h. Huh, and K. Iwatsuki. 2002. Dynamic alteration of human β-defensin 2 localization from cytoplasm to intercellular space in psoriatic skin. Journal of molecular medicine. 80:678-684.
2001
Kameda, T., S. Hatakeyama, K. Terada, and T. Sugiyama. 2001. Acceleration of the formation of cultured epithelium using the sonic hedgehog expressing feeder cells. Tissue engineering. 7:545-555.