Instructions HSkMC Normal
5 Important Cell Culture Rules
MSDS Cryopreserved Cells
Cell Apps Flyer Skeletal Muscle Cells
Cell Apps Poster Primary Cells
Cell Applications Inc Brochure
Description
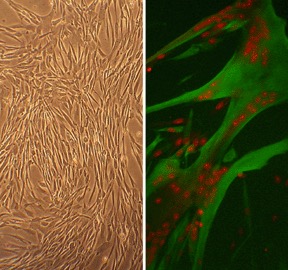
Human Skeletal Muscle Cells (HSkMC), isolated from adult or fetal donor limbs are positive for sarcomere myosin and can undergo differentiation to exhibit actin and myosin myofilaments. Our HSkMC provide a useful model system to study many aspects of muscular function and disease. Skeletal Muscle Cells play an instrumental role in the glucose metabolism and diabetes.
Select HSkMC lots have been additionally tested to demonstrate stimulation-dependent AMPK signaling and responsiveness to insulin stimulation. More information about pre-screened skeletal muscle cells can be found on the Pre-Screened Skeletal Muscle Cell Product Page.
HSkMC from Cell Applications, Inc. have been used to:
- Serve as a differentiated control in a study of developmental regulator genes in hESC
- Characterize statin-induced gene expression changes and demonstrate cytotoxic effect of statins in skeletal muscle cells
- Identify molecular mechanisms of mitochondrial myopathy and sideroblastic anemia resulting from a missense mutation in the PUS1 gene
- Investigate chemokine-like factor expression in the idiopathic inflammatory myopathies
- Demonstrate that fasting activates AceCS2 gene expression by inducing KLF15 transcription factor
- Characterize human FGFR3-positive sarcoma-initiating stem cells
- Identify trans-Golgi network proteins and Notch and Hedgehog pathways as putative targets for rhabdomyosarcoma therapy
- Show that riluzole muscle relaxant effects are mediated by inhibition of INa and stimulation of BKC-channel activity
- Develop nitric esters that combine the pharmacological functions of NO and muscle relaxation properties for treatment of muscular diseases
- Develop biodegradable polymer-based transgene delivery vectors for muscular dystrophy treatment
- Design optimal coating for orthopedic metallic implants
Details
Tissue | Normal healthy human limb skeletal muscle |
QC | No bacteria, yeast, fungi, mycoplasma, virus |
Character | Multinucleated myotubes form after culture in fusion medium |
Bioassay | Attach, spread, proliferate in Growth Med |
Cryovial | 500,000 HSkMC (2nd passage) frozen in Basal Medium w/ 10% FBS, 10% DMSO |
Kit | Cryovial frozen HSkMC (150-05), Growth Medium (151-500), Subcltr Rgnt Kit (090K) |
Proliferating | Shipped in Tsfr Med, psg 3, flasks or plates |
Doublings | At least 15 |
Applications | Laboratory research use only (RUO). Not for human, clinical, diagnostic or veterinary use. |
Products
| Product | Size | CAT.# | Price | Quantity |
|---|---|---|---|---|
| Cryopreserved Human Skeletal Muscle Cell Total Kit, fetal: 5x10^5 Cells (Fetal), Medium & Subculture Reagents (See Details tab for specifics) | Size: 1 Kit | CAT.#: 150K-05f | Price: $925.00 | |
| Cryopreserved Human Skeletal Muscle Cell Total Kit, adult: 5x10^5 Cells (Adult), Medium & Subculture Reagents (See Details tab for specifics) | Size: 1 Kit | CAT.#: 150K-05a | Price: $925.00 | |
| Cryopreserved Pre-Screened Skeletal Muscle Cells Total Kit, fetal: 5x10^5 Cells (Pre-Screened, Fetal), Medium & Subculture Reagents (See Details tab for specifics) | Size: 1 Kit | CAT.#: S150K-05f | Price: $1,045.00 | |
| Cryopreserved Skeletal Muscle Cells (HSkMC), adult: Frozen HSkMC (5x10^5) | Size: 1 Cryovial | CAT.#: 150-05a | Price: $725.00 | |
| Cryopreserved Skeletal Muscle Cells (HSkMC), fetal: Frozen HSkMC (5x10^5) | Size: 1 Cryovial | CAT.#: 150-05f | Price: $725.00 | |
| Cryopreserved Skeletal Muscle Cells (HSkMC), Pre-Screened, fetal: Frozen HSkMC (5x10^5) | Size: 1 Cryovial | CAT.#: S150-05f | Price: $845.00 | |
| Cryopreserved Pre-Screened Skeletal Muscle Cells with COPD Total Kit, adult: 5x10^5 Cells (from donor with COPD, Pre-Screened, Adult), Medium & Subculture Reagents (See Details tab for specifics) | Size: 1 Kit | CAT.#: S150COPDK-05a | Price: $1,175.00 | |
| Cryopreserved Skeletal Muscle Cells, COPD (HSkMC-COPD), Pre-Screened, adult: Frozen HSkMC-COPD from donor with Chronic Obstructive Pulmonary Disease, Adult (5x10^5) | Size: 1 Cryovial | CAT.#: S150COPD-05a | Price: $975.00 | |
| Cryopreserved Human Skeletal Muscle Type-2 Diabetes Total Kit, adult: 5x10^5 Cells (from donor with Type 2 Diabetes, Adult), Medium & Subculture Reagents (See Details tab for specifics) | Size: 1 Kit | CAT.#: 150T2DK-05a | Price: $1,025.00 | |
| Cryopreserved Skeletal Muscle Cells, Type 2 Diabetes (HSkMC-T2D), adult: Frozen HSkMC from donor with Type 2 Diabetes (5x10^5) | Size: 1 Cryovial | CAT.#: 150T2D-05a | Price: $825.00 | |
| Proliferating Skeletal Muscle Cells (HSkMC), adult: Actively growing, dividing cells in medium | Size: T-25 Flask | CAT.#: 151-25a | Price: $725.00 | |
| Proliferating Skeletal Muscle Cells (HSkMC), adult: Actively growing, dividing cells in medium | Size: T-75 Flask | CAT.#: 151-75a | Price: $915.00 | |
| Proliferating Skeletal Muscle Cells (HSkMC), adult: Actively growing, dividing cells in medium | Size: 24 Well | CAT.#: 151-24Wa | Price: $915.00 | |
| Proliferating Skeletal Muscle Cells (HSkMC), adult: Actively growing, dividing cells in medium | Size: 96 Well | CAT.#: 151-96Wa | Price: $1,035.00 | |
| Proliferating Skeletal Muscle Cells (HSkMC), fetal: Actively growing, dividing cells in medium | Size: T-25 Flask | CAT.#: 151-25f | Price: $725.00 | |
| Proliferating Skeletal Muscle Cells (HSkMC), fetal: Actively growing, dividing cells in medium | Size: T-75 Flask | CAT.#: 151-75f | Price: $915.00 | |
| Proliferating Skeletal Muscle Cells (HSkMC), fetal: Actively growing, dividing cells in medium | Size: 24 Well | CAT.#: 151-24Wf | Price: $915.00 | |
| Proliferating Skeletal Muscle Cells (HSkMC), fetal: Actively growing, dividing cells in medium | Size: 96 Well | CAT.#: 151-96Wf | Price: $1,035.00 | |
| Proliferating Skeletal Muscle Cells (S-HSkMC), Pre-Screened, fetal: Actively growing, dividing cells in medium | Size: T-25 Flask | CAT.#: S151-25f | Price: $845.00 | |
| Proliferating Skeletal Muscle Cells (S-HSkMC), Pre-Screened, fetal: Actively growing, dividing cells in medium | Size: T-75 Flask | CAT.#: S151-75f | Price: $1,035.00 | |
| Proliferating HSkMC, Pre-Screened, Fetal: Actively growing, dividing cells in medium | Size: 24 Well | CAT.#: S151-24Wf | Price: $990.00 | |
| Proliferating Skeletal Muscle Cells (S-HSkMC), Pre-Screened, fetal: Actively growing, dividing cells in medium | Size: 96 Well | CAT.#: S151-96Wf | Price: $1,155.00 |
Related Products
| Product | Size | CAT.# | Price | Quantity |
|---|---|---|---|---|
| HSkMC Growth Medium: All-in-one ready-to-use | Size: 500 ml | CAT.#: 151-500 | Price: $147.00 | |
| HSkMC Growth Medium Kit: Basal medium & growth supplement sold together packaged separately | Size: Yields 500 ml | CAT.#: 151K-500 | Price: $147.00 | |
| HSkMC Basal Medium: Basal medium (contains no growth supplement). Add GS before use. | Size: 500 ml | CAT.#: 150-500 | Price: $81.00 | |
| HSkMC Growth Supplement: Added to Basal Medium to create Growth Medium | Size: 50 ml (2 parts) | CAT.#: 151-GS | Price: $85.00 | |
| HSkMC Differentiation Medium: Promotes cells to change from one type to another, more specialized | Size: 250 ml | CAT.#: 151D-250 | Price: $81.00 |
Extended Family Products
| Product | Size | CAT.# | Price | Quantity |
|---|---|---|---|---|
| Mouse Interleukin-6 Antibody: Mouse Interleukin-6 Antibody | Size: 100 ul | CAT.#: CP10325 | Price: $302.00 | |
| Freezing Medium: For general cryopreservation of most primary cells. Contains FBS & DMSO. | Size: 50 ml | CAT.#: 040-50 | Price: $54.00 | |
| Cyto-X Cell Counting Reagent: 500 tests | Size: 1 Bottle | CAT.#: 028-01 | Price: $160.00 | |
| Cyto-X Cell Counting Reagent Sample: 100 tests | Size: Sample | CAT.#: 028-S | Price: $41.00 | |
| Skeletal Muscle Cell RNA (HSkMC RNA), Adult: Total RNA prepared from Human Skeletal Muscle Cells, adult | Size: 10 ug | CAT.#: 150-R10a | Price: $398.00 | |
| Skeletal Muscle Cell RNA (HSkMC RNA), Adult: Total RNA prepared from Human Skeletal Muscle Cells, adult | Size: 25 ug | CAT.#: 150-R25a | Price: $796.00 | |
| Skeletal Muscle Cell RNA (HSkMC RNA), Fetal: Total RNA prepared from Human Skeletal Muscle Cells, fetal | Size: 10 ug | CAT.#: 150-R10f | Price: $398.00 | |
| Skeletal Muscle Cell RNA (HSkMC RNA), Fetal: Total RNA prepared from Human Skeletal Muscle Cells, fetal | Size: 25 ug | CAT.#: 150-R25f | Price: $796.00 | |
| Skeletal Muscle Cell RNA (HSkMC RNA), Multinucleated Myotubes, Fetal: Total RNA prepared from Human Skeletal Muscle Cells, Differentiated, fetal | Size: 10 ug | CAT.#: 150D-R10f | Price: $514.00 | |
| Skeletal Muscle Cell RNA (HSkMC RNA), Multinucleated Myotubes, Fetal: Total RNA prepared from Human Skeletal Muscle Cells, Differentiated, fetal | Size: 25 ug | CAT.#: 150D-R25f | Price: $1,028.00 | |
| Human Interleukin-6 (IL-6): Human Interleukin-6 | Size: 20 ug | CAT.#: RP1010-20 | Price: $194.00 | |
| Human Interleukin-6 (IL-6): Human Interleukin-6 | Size: 100 ug | CAT.#: RP1010-100 | Price: $484.00 | |
| Human Interleukin-6 (IL-6): Human Interleukin-6 | Size: 1000 ug | CAT.#: RP1010-1000 | Price: $3,175.00 | |
| Human IL-6 ELISA Kit: Human Interleukin-6 ELISA Kit | Size: 96 Wells | CAT.#: CL0410 | Price: $517.00 | |
| Human Skeletal Muscle RNA: Total RNA prepared from human skeletal muscle tissue | Size: 50 ug | CAT.#: 1H60-50 | Price: $158.00 | |
| Human Skeletal Muscle RNA: Total RNA prepared from human skeletal muscle tissue | Size: 250 ug | CAT.#: 1H60-250 | Price: $595.00 | |
| Subculture Reagent Kit: 100 ml each of HBSS, Trypsin/EDTA & Trypsin Neutralizing Solution | Size: 1 Kit | CAT.#: 090K | Price: $63.00 | |
| Mouse Myostatin Antibody: Mouse Myostatin Antibody | Size: 100 ul | CAT.#: CP10315 | Price: $302.00 | |
| Human Myostatin (GDF-8): Human Myostatin | Size: 10 ug | CAT.#: RP1022-10 | Price: $194.00 | |
| Human Myostatin (GDF-8): Human Myostatin | Size: 100 ug | CAT.#: RP1022-100 | Price: $624.00 | |
| Human Myostatin (GDF-8): Human Myostatin | Size: 1000 ug | CAT.#: RP1022-1000 | Price: $5,327.00 | |
| Human Myostatin Propeptide: Human Myostatin Propeptide | Size: 25 ug | CAT.#: RP1142-25 | Price: $194.00 | |
| Human Myostatin Propeptide: Human Myostatin Propeptide | Size: 100 ug | CAT.#: RP1142-100 | Price: $624.00 | |
| Human Myostatin Propeptide: Human Myostatin Propeptide | Size: 1000 ug | CAT.#: RP1142-1000 | Price: $5,327.00 | |
| Human IL-6, Animal Free: Human Interleukin-6, Animal-Free | Size: 20 ug | CAT.#: RP1010AF-20 | Price: $213.00 | |
| Human IL-6, Animal Free: Human Interleukin-6, Animal-Free | Size: 100 ug | CAT.#: RP1010AF-100 | Price: $533.00 | |
| Human IL-6, Animal Free: Human Interleukin-6, Animal-Free | Size: 1000 ug | CAT.#: RP1010AF-1000 | Price: $3,492.00 | |
| HSkMC Starvation Medium: Use when cells need to be starved overnight to 24 hrs before experiment | Size: 250 ml | CAT.#: 151S-250 | Price: $71.00 |
Resources/Documents
Publications
2016
Duncan, D., T. Kamerzell and M. Palmer. 2016. Use of Indole Compounds for Fat Reduction and Skin and Soft Tissue Tightening. Patent Applications US 20160030389 A1.
2014
Iizuka, K., T. Machida, and M. Hirafuji. 2014. Extracellular MCT4 Is a Possible Indicator for Skeletal Muscle MHC Fiber Type Change. Ann Clin Lab Sci, 44:272-276.
2013
Wang, G., and Q. Lu. 2013. A nitrate ester of sedative alkyl alcohol improves muscle function and structure in a murine model of Duchenne muscular dystrophy. Molecular pharmaceutics. 10:3862-3870.
2012
Idris, N., M. Ashraf, R. Ahmed, J. Shujia, and K. Haider. 2012. Activation of IL-11/STAT3 pathway in preconditioned human skeletal myoblasts blocks apoptotic cascade under oxidant stress. Regen Med, 7:47-57.
Nagao, H., T. Setoguchi, S. Kitamoto, Y. Ishidou, S. Nagano, M. Yokouchi, M. Abematsu, N. Kawabata, S. Maeda, S. Yonezawa, and S. Komiya. 2012. RBPJ Is a Novel Target for Rhabdomyosarcoma Therapy. PloS one. 7:e39268.
Wang, M., J.D. Tucker, P. Lu, B. Wu, C. Cloer, and Q. Lu. 2012. Tris[2-(acryloyloxy)ethyl]isocyanurate Cross-Linked Low-Molecular-Weight Polyethylenimine as Gene Delivery Carriers in Cell Culture and Dystrophic mdx Mice. Bioconjugate chemistry. 23:837-845.
2011
Kawabata, N., K. Ijiri, Y. Ishidou, T. Yamamoto, H. Nagao, S. Nagano, S. Maeda, S. Komiya, and T. Setoguchi. 2011. Pharmacological inhibition of the Hedgehog pathway prevents human rhabdomyosarcoma cell growth. International journal of oncology. 39:899.
Kunigou, O., H. Nagao, N. Kawabata, Y. Ishidou, S. Nagano, S. Maeda, S. Komiya, and T. Setoguchi. 2011. Role of GOLPH3 and GOLPH3L in the proliferation of human rhabdomyosarcoma. Oncology reports. 26:1337-1342.
2010
Thuangtong, R., J.J. Bentow, K. Knopp, N.A. Mahmood, N.E. David, and M.S. Kolodney. 2010. Tissue‐Selective Effects of Injected Deoxycholate. Dermatologic Surgery. 36:899-908.
Zalevsky, J., D. Nguyen, G. Moore, S. Ezhevsky, J. Desjarlais, A. Chirino, D. Cash, and M. Bernett. 2010. Pharmaceutical compositions of adiponectin variants and methods of storage. Patent US 7678886 B2.
Zalevsky, J., D. Nguyen, G. Moore, S. Ezhevsky, J. Desjarlais, A. Chirino, D. Cash, and M. Bernett. 2010. Adiponectin variants. Patent US 7709607 B2.
Zalevsky, J., D. Nguyen, G. Moore, S. Ezhevsky, J. Desjarlais, A. Chirino, D. Cash, and M. Bernett. 2010. Method of treatment using adiponectin variants. Patent US 7749956 B2.
2009
Gupta, A., C. Lobocki, S. Singh, M. Robertson, O.A. Akadiri, G. Malhotra, and I.T. Jackson. 2009. Actions and Comparative Efficacy of Phosphatidylcholine Formulation and Isolated Sodium Deoxycholate for Different Cell Types. Aesth Plast Surg. 33:346-352.
Hirotsu, M., T. Setoguchi, Y. Matsunoshita, H. Sasaki, H. Nagao, H. Gao, K. Sugimura, and S. Komiya. 2009. Tumour formation by single fibroblast growth factor receptor 3-positive rhabdomyosarcoma-initiating cells. Br J Cancer. 101:2030-2037.
Peters, L., and T. Lorenz. 2009. Postsynaptically Targeted Chemodenervation Agents and Their Methods of Use. Patent Application US 20110118190 A1.
Xu, J., S. Ekins, M. McGlashen, and D. Lauffenburger. 2009. FUTURE PERSPECTIVES OF BIOLOGICAL ENGINEERING IN PHARMACEUTICAL RESEARCH: THE PARADIGM OF MODELING, MINING, MANIPULATION, AND MEASUREMENTS. Ch. 14 inXu, J. and Ekins, S. 2009. Drug Efficacy, Safety, and Biologics Discovery: Emerging Technologies and Tools.
Xu, J. and L. Yu. 2009. Focus on the Fundamentals: Toward Better Therapeutic Index Prediction. Pg 19. In: Drug Efficacy, Safety, and Biologics Discovery: Emerging Technologies and Tools. Ekins S. and X Jinghai, Eds. John Wiley & Sons, Inc.
Zalevsky, J., D. Nguyen, G. Moore, S. Ezhevsky, J. Desjarlais, A. Chirino, D. Cash, and M. Bernett. 2009. Globular adiponectin variants. Patent US 7592423 B2.
2008
Wang, Y.-J., M.-W. Lin, A.-A. Lin, and S.-N. Wu. 2008. Riluzole-induced block of voltage-gated Na+ current and activation of BKCa channels in cultured differentiated human skeletal muscle cells. Life sciences. 82:11-20.
2006
Cash, D., A. Chirino, J. Desjarlais, S. Ezhevsky, G. Moore, D. Nguyen, and J. Zalevsky. 2006. Adiponectin variants. Patent Application US 20070015909 A1.
Perla, V., and T.J. Webster. 2006. nano-hydroxyapatite–thermally denatured small intestine sub-mucosa composites for entheses applications. International Journal of nanomedicine. 1:351.
Yamazaki, H., M. Suzuki, T. Aoki, S. Morikawa, T. Maejima, F. Sato, K. Sawanobori, M. Kitahara, T. Kodama, and Y. Saito. 2006. Influence of 3-hydroxy-3-methylglutaryl coenzyme A reductase inhibitors on ubiquinone levels in rat skeletal muscle and heart: relationship to cytotoxicity and inhibitory activity for cholesterol synthesis in human skeletal muscle cells. Journal of atherosclerosis and thrombosis. 13:295-307.
Zalevsky, J., D. Nguyen, G. Moore, S. Ezhevsky, J. Desjarlais, A. Chirino, D. Cash, and M. Bernett. 2006. Rational Chemical Modification of Adiponectin Variants. Patent Application US 20070054359 A1.
2005
Morikawa, S., T. Murakami, H. Yamazaki, A. Izumi, Y. Saito, T. Hamakubo, and T. Kodama. 2005. Analysis of the global RNA expression profiles of skeletal muscle cells treated with statins. J. atherosclerosis & thrombosis. 12:121-131.
Patton, J.R., Y. Bykhovskaya, E. Mengesha, C. Bertolotto, and N. Fischel-Ghodsian. 2005. Mitochondrial Myopathy and Sideroblastic Anemia (MLASA): Missense mutation in the pseudouridine synthase 1 (PUS1) gene is associated with the loss of tRNA pseudouridylation. J. Biological Chemistry. 280:19823-19828.
Perla, V., M. Sato, and T.J. Webster. 2005. Increased Skeletal Muscle Cell and Osteoblast Numbers on Hydrothermally-Treated Nano-Hydroxyapatite/Collagen Type I Composites for Entheses Applications. Journal of Biomedical Nanotechnology. 1:297-305.
2004
Yamamoto, J., Y. Ikeda, H. Iguchi, T. Fujino, T. Tanaka, H. Asaba, S. Iwasaki, R.X. Ioka, I.W. Kaneko, K. Magoori, S. Takahashi, T. Mori, H. Sakaue, T. Kodama, M. Yanagisawa, T.T. Yamamoto, S. Ito, and J. Sakai. 2004. A Krüppel-like factor KLF15 Contributes Fasting-induced Transcriptional Activation of Mitochondrial Acetyl-CoA Synthetase Gene AceCS2. Journal of Biological Chemistry. 279:16954-16962.