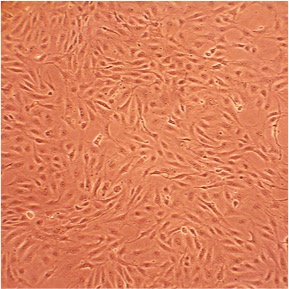
Porcine Aortic Endothelial Cells: PAOEC
Description
Porcine Aortic Endothelial Cells (PAOEC) provide a useful model system to study many aspects of cardiovascular function and disease. Co-culture of the artery endothelial cells with species-matched smooth muscle cells provides an ideal model for studying the interaction between these two cell types.
PAOEC from Cell Applications, Inc., and they have been utilized in a number of research publications, including those demonstrating that:
- H2O2 contributes to vascular dysfunction
- Cell survival under oxidative stress depends on phosphorylation of VEGFR-3
- Replication of swine fever virus regulates signal transduction pathways and gene expression
- Direct interactions between endothelial cells and T cells trigger release of proinflammatory molecules that play a role in graft rejection
- The tetraspanin CD82 is the recognition sensor responsible for rejection of xenotransplants
- Endothelial cells from heart valves align perpendicular to flow, while aortic endothelial cells align parallel to flow, indicating the need to match the cell types when designing engineered tissue devices
- Shear stress induces changes in expression of NO synthase, VCAM-1, c-jun, MCP-1 and ICAM-1
- Swine-to-human xenotransplantation can be improved by pretreating donors with vasopressin
- Endothelial implants can be designed to increase lumen diameter and replace heart valves
Details
| Tissue | Normal healthy porcine aorta | |
|---|---|---|
| QC | No bacteria, yeast, fungi, mycoplasma | |
| Character | DiI-Ac-LDL uptake: Positive | |
| Bioassay | Attach, spread, proliferate in Growth Med | |
| Cryovial | 500,000 PAOEC (1st passage) frozen in Basal Medium w/ 10% FBS, 10% DMSO | |
| Kit | Cryovial frozen PAOEC, Growth Medium (P211-500), Subculture Rgnt Kit (090K) | |
| Proliferating | Shipped in Gr Med, 2nd psg (flasks or plates) | |
| Doublings | At least 16 | |
| Applications | Laboratory research use only (RUO). Not for human, clinical, diagnostic or veterinary use. |
Resources
FAQs
Need More Help?
Visit our comprehensive FAQ page for detailed answers to common questions.
Need More Help?
Visit our comprehensive FAQ page for detailed answers to common questions.
Primary Cell FAQs