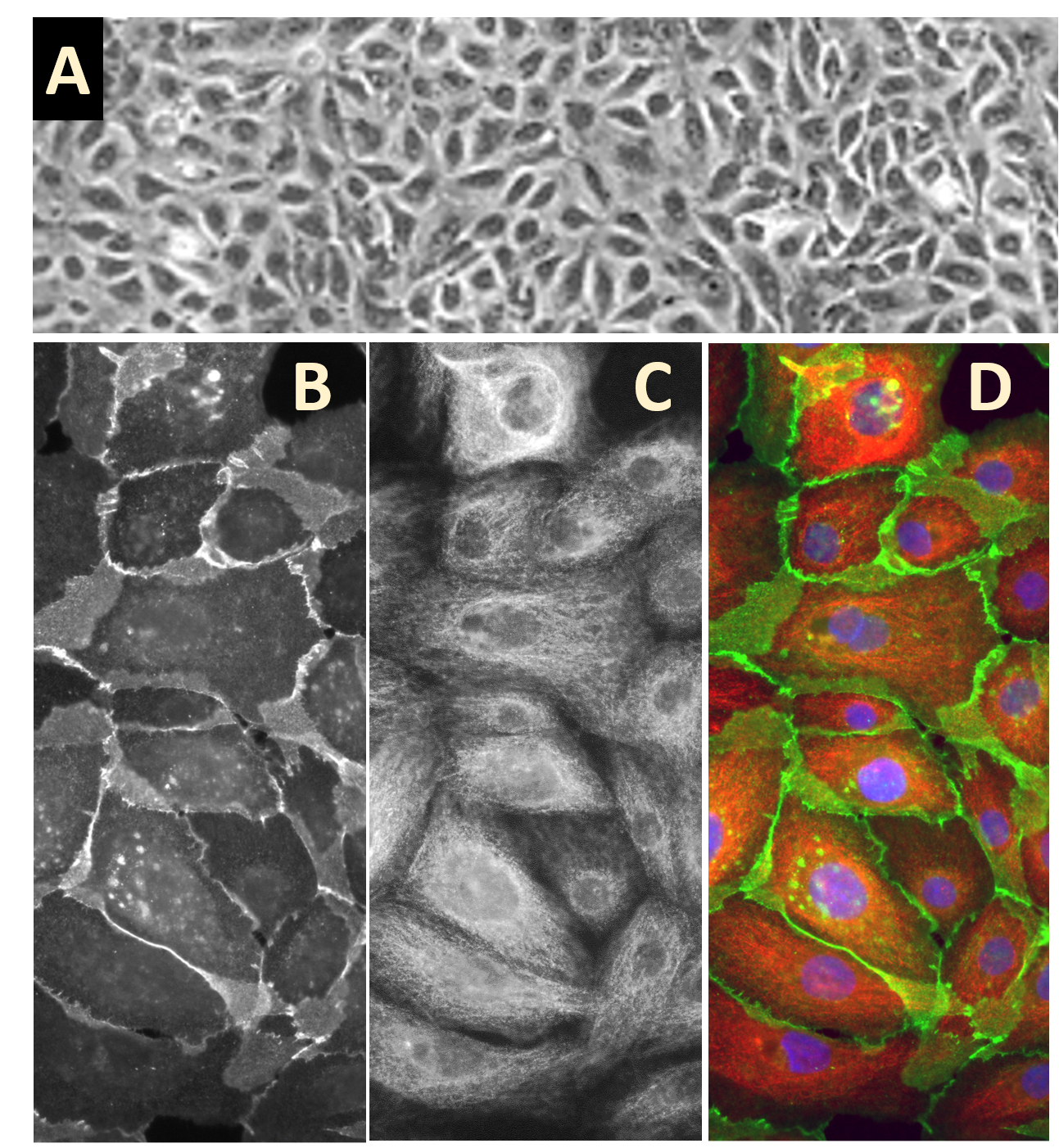
Human Mammary Epithelial Cells: HMEpC
Description
Human Mammary Epithelial Cells (HMEpC) provide an excellent model system to study many aspects of epithelial function and disease, particularly those related to cancerogenesis.
HMEpC from Cell Applications, Inc. have been utilized in numerous research publications, for example to:
- Investigate the role of exosomes secreted by cancer cells in formation of tumor permissive microenvironment through manipulation of normal mammary epithelium
- Serve as control in a study investigating antitumor properties of cannabinoids and stem cell microenvironment
- Determine that differential expression of glycoproteins allows classification of human breast cells into normal, benign, malignant, basal, and luminal groups
- Identify ALDH isoform 5A1 as a potential target for treatment of human breast ductal carcinoma
- Determine that combination of an anti-growth factor treatment using phototherapy (UV-B) is more effective
- Investigate cell survival following DNA damage
- Show the important roles of tumor suppressors in mammary epithelial differentiation
- Explain protection of extracellular matrix from degradation in normal mammary epithelia,
- Show that Maspin loss in metastatic cancer leads to unrestricted ECM degradation, contributing to metastasis, and that loss of EcSOD expression also promotes invasiveness by disrupting ECM
- Investigate the role of shortened telomeres in initiation of genomic instability, cytokinesis failure and polyploidy
- Elucidate the role of Myc in malignancy by studying its ability to transform primary epithelial cells
- Demonstrate, along with Human dermal Fibroblasts, also from Cell Applications, Inc., that resveratrol inhibits mono-ubiquitination of histone H2B
Characterization: Morphology consistent with epithelial origin, and positive for epithelial cell marker cytokeratin 18
Details
| Tissue | Normal healthy human mammary glands | |
|---|---|---|
| QC | No bacteria, yeast, fungi, mycoplasma, virus | |
| Bioassay | Attach, spread, proliferate in Growth med | |
| Cryovial | 500,000 HMEpC (5th passage) frozen in Basal Medium w/ 10% FBS, 10% DMSO | |
| Kit | Cryovial frozen HMEpC(830-05a), Growth Med (815-500), Subculture Rgnt Kit (090K) | |
| Proliferating | Shipped in Gr Med, 6th psg (flasks or plates) | |
| Doublings: | At least 16 | |
| Applications | Laboratory research use only (RUO). Not for human, clinical, diagnostic or veterinary use. |
Resources
FAQs
Need More Help?
Visit our comprehensive FAQ page for detailed answers to common questions.
Need More Help?
Visit our comprehensive FAQ page for detailed answers to common questions.
Primary Cell FAQs