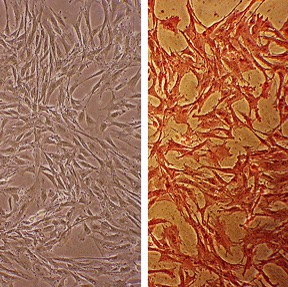
Bovine Aortic Smooth Muscle Cells: BAOSMC
Description
Bovine Aortic Smooth Muscle Cells (BAOSMC) provide an excellent model system to study all aspects of cardiovascular function and disease, especially those related to mechanisms of hyperplasia and hypertrophy of intimal smooth muscle cells leading to vascular occlusion in atherosclerosis and stent restenosis.
BAOSMC from Cell Applications, Inc. have been utilized in a number of research studies, for example, to:
- Elucidate RhoA-dependent serum response signaling pathways
- Observe the role of FAK in thrombospondin-1 induced migration of vascular smooth muscle cells
- Demonstrate the relationship between the disturbed flow and proliferation of smooth muscle cells, leading to intima hyperplasia
- Study interactions between lipoproteins and extracellular matrix, as well as mechanisms of lipoprotein aggregation contributing to atherosclerosis
- Construct an expression cassette to maximize targeted transgene expression
- Test new technologies, such as on-chip contact imaging technique, handheld fluorometers and a “nose on a chip” olfactory sensor
- Study the behavior of smooth muscle cells in 3d environment and smooth muscle/endothelial co-cultures
- Describe an origami-like method of generating three-dimensional (3D) cell-laden microstructures
- Design materials for tissue engineering and cardiovascular implants
Characterization: positive for smooth muscle cell specific alpha-actin expression
Details
| Tissue | Normal healthy bovine aorta | |
|---|---|---|
| QC | No bacteria, yeast, fungi, mycoplasma | |
| Character | Smooth muscle specific α-actin positive | |
| Bioassay | Attach, spread, proliferate in Growth Med | |
| Cryovial | 500,000 BAOSMC (2nd passage) frozen in Basal Medium w/ 10% FBS, 10% DMSO | |
| Kit | Cryovial frozen BAOSMC(B354-05), Growth Medium (311-500), Subcltr Rgnt Kit (090K) | |
| Proliferating | Shipped in Gr Med, 3rd psg (flasks or plates) | |
| Doublings | At least 16 | |
| Applications | Laboratory research use only (RUO). Not for human, clinical, diagnostic or veterinary use. |
Resources
FAQs
Need More Help?
Visit our comprehensive FAQ page for detailed answers to common questions.
Need More Help?
Visit our comprehensive FAQ page for detailed answers to common questions.
Primary Cell FAQs